Haematopoietic progenitor cells (HPC)
Why measure haematopoietic progenitor cells?
Now anyone in the laboratory can do a stem cell count at any time using the automated measurement option ‘XN Stem Cells’ to quantify haematopoietic progenitor cells on the XN-Series WPC measurement channel.
Simple, fast and reliable, the HPC count is available within a few minutes as there is no need for manual gating, pre-treatment or sample washing as compared to immune flow cytometry measurement. There is an excellent correlation with the CD34 immune flow cytometry. XN Stem Cells is a functional equivalent of CD34 analysis and may be a surrogate for CD34 analysis to predict the optimal timing of stem cell collections from mobilised peripheral blood.
Having a faster count available supports tighter monitoring of the mobilisation process for accurately determining the optimal timing for starting apheresis. The intermediate product can now be analysed quickly and reliably during apheresis to monitor the stem cell yield and optimise the collection time. This strongly reduces uncertainties as to the best time to start and stop collecting stem cells.
Thanks to the optimised apheresis workflow, you can expect an overall reduction in costs. You will also enjoy a higher turnaround of patients and better resource management on the apheresis ward since patients may be accepted for apheresis earlier or do not get turned down. Finally, the patient experience will improve in most cases as some patients can avoid repeated apheresis while others may leave earlier, with all of them knowing that collection has been successful.
Where to use HPC
XN Stem Cells offers a fast method for monitoring both stem cell mobilisation and collection, and predicts product quality. Hence all hospitals performing haematopoietic stem cell transplantations can now benefit from the HPC count delivered by the XN-Series. In addition, all specialised apheresis wards performing stem cell apheresis can benefit directly from enumerating stem cells from the intermediate product during collection.
How are HPC measured?
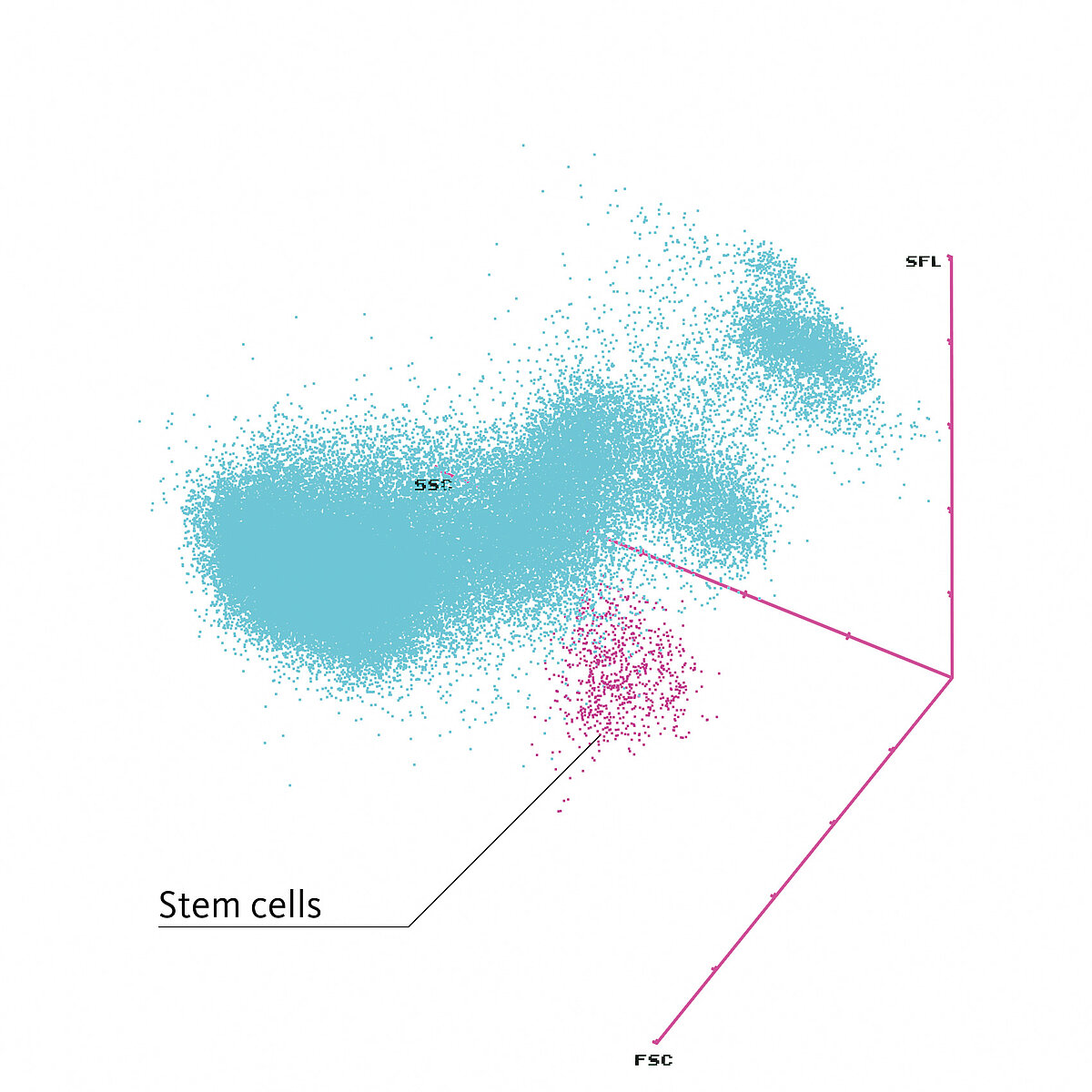
With its unique combination of reagents, the WPC channel detects abnormal membrane composition and nuclear content. The lipid membrane composition of immature cells is different from that of mature cells or abnormal blasts. Stem cells’ membranes are relatively resistant to permeabilisation by the WPC reagent. As a result, stem cells are medium in size (medium FSC), have a low granularity (low SSC) and relatively low fluorescence intensity (low-medium SFL).