A new technique for melanoma treatment
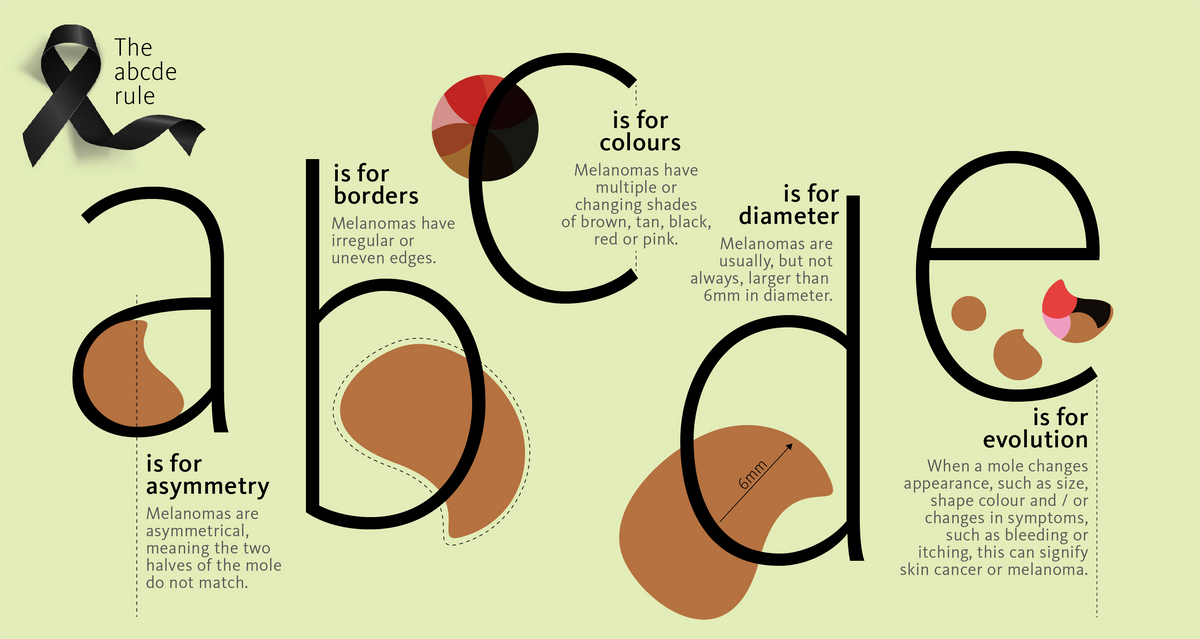
Melanoma and skin cancer
Worldwide, incidences of skin cancer are increasing. While there are several types of skin cancer, melanoma is the deadliest, killing around 50,000 people worldwide each year [1]. The good news is that the range of treatment options is expanding, and if diagnosed early, patients have a much higher likelihood of beating the disease.
Protect yourself with small, everyday changes
We all know that too much sun exposure can be dangerous – most of us have experienced the pain of a sunburn. Even in conditions that we don’t expect sun damage to occur, such as cloudy days or when it’s cold outside, it’s still possible.
Melanoma is caused by skill cells that begin to develop abnormally, which exposure to ultraviolet (UV) light is thought to cause. Sudden, intense sun exposure can be the most unexpected, but also the most dangerous. That’s why it’s important to be informed and prepared.
Why the focus on melanoma?
Melanoma is much less common than some of the other types of skin cancer, but melanoma is more dangerous because it more likely to spread quickly than other types.
Sun exposure adds up over time, so it’s logical that is more likely to occur in older people. However, melanoma is one of the most common cancers in people younger than 30, making it extremely important to educate younger generations of the dangers of skin cancer.
Treatment solutions in melanoma - precisely bring to light what needs to be seen
Melanoma is often considered one of the most aggressive and treatment-resistant human cancers [4]. In the past few years, the therapeutic landscape of melanoma has been revolutionised by immunotherapies and targeted therapies. Both strategies have shown markedly improved survival compared with the use of chemotherapy regimens [5].
Precision diagnostics are key to selecting the right therapy at the right time for the right cancer patient.
Gene alterations play a key role during the onset and development of melanoma [6]. Therefore, reliable assessment of the tumour’s mutational status is decisive not only in selecting cancer patients eligible for targeted therapy, but also later on to monitor response to treatment and disease progression.
To investigate such genetic changes, oncologists can make use of a variety of molecular genetic tests which may support them in making personalised treatment decisions.
Sysmex offers a range of products based on cutting-edge molecular biology technologies supporting heath care professionals and patients along the whole pathway of care.
Molecular tests for therapy selection
CytoCell FISH (Fluorescent In Situ Hybridization) probes targeting genes in chromosomes 6 and 11 (RREB1, MYB and CCND1) are used to aid pathologists and clinicians in differentiating benign versus malignant melanocytic lesions.
BRAF, NRAS, KIT and NF1 are among the most common mutated genes underlying the pathogenesis of melanoma that can be investigated by next generation sequencing (NGS) tests. The Sysmex solution to help characterise the overall genomic landscape of melanomas is SureSeq myPanelTM* NGS Custom Melanoma Cancer Panel.
For first-line treatment particularly in BRAF wild-type patients, immunotherapy with PD-1 antibodies alone or in combination with CTLA-4 antibodies should be considered [7]. While the combination of dabrafenib/trametinib is one of the standards of care for adjuvant BRAF-mutated melanoma patients and was approved by the European Medicines Agency for adjuvant treatment for melanoma in August 2018 [5].
Liquid biopsy for monitoring
Signal transduction inhibition and immune checkpoint inhibition have greatly improved the treatment of patients with metastatic melanoma. Unfortunately, not all patients respond to such treatment. The standard way to measure whether the treatment is working is through radiological assessment. However, it may be too late to help the patient by the time it indicates resistance, which increases the need to find better ways to predict patient response to treatment.
Liquid biopsy analysis can offer essential and timely information that shows how the tumour is responding to the treatment and detect minimal residual disease. Through a simple and minimally invasive blood draw, liquid biopsy can reveal clinically relevant mutations in circulating-tumour DNA (ctDNA) that the tumour cells shed into the bloodstream. This information allows clinicians to monitor therapy in real time by repeated sampling, detect progression earlier than routine radiological scans and switch therapy if needed. [14]
Plasma-SeqSensei™ IVD Solid Cancer Kit enables highly sensitive and quantitative liquid biopsy analysis and detection on relevant mutations in BRAF, EGFR, KRAS, NRAS and PIK3CA genes utilising next-generation sequencing technology. PSS's short and standardised workflow delivers results within two days including the generation of easy-to-read reports using laptop-compatible Plasma-SeqSensei™ IVD Software.
Find out more about how Sysmex makes a difference in the battle to conquer melanoma.
References
[1] Cancer Incidence and Mortality Worldwide: IARC CancerBase http://globocan.iarc.fr/Pages/fact_sheets_population.aspx
[2] Piñero-Madrona et al. (2020): J Surg Oncol. 1-6
[3] Anninga et al. (2016). (2016): Ann Surg Oncol. 23(6):2070-8
[4] Tsao et al, Genes & Dev. 2012. 26: 1131-1155
[5] O. Michielin et al. Cutaneous melanoma: ESMO Clinical Practice (2019) Annals of Oncology 30: 1884–1901
Guidelines for diagnosis, treatment and follow-up
[6] Sini M.C., Doneddu V., Paliogiannis P., Casula M., Colombino M., Manca A., Botti G., Ascierto P.A., Lissia A., Cossu A. Palmieri G. Genetic alterations in main candidate genes during melanoma progression. Oncotarget 9: 8531-8541, 2018
[7] Garbe et al, European Journal of Cancer 63 (2016) 201-217
[8] Overman MJ, Modak J, Kopetz S, et al: Use of research biopsies in clinical trials: Are risks and benefits adequately discussed? J Clin Oncol 31:17-22, 2013
[9] Vogelstein B, Papadopoulos N, Velculescu VE, et al: Cancer genome landscapes. Science 339:1546-1558, 2013
[10] Gerlinger M, Rowan AJ, Horswell S, et al: Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N Engl J Med 366:883-892, 2012
[11] https://www.cancer.org/cancer/melanoma-skin-cancer/causes-risks-prevention/risk-factors.html
[12] https://www.cancer.org/cancer/melanoma-skin-cancer/about/what-is-melanoma.html
[13] https://www.nhs.uk/conditions/melanoma-skin-cancer/#:~:text=Melanoma%20is%20caused%20by%20skin,melanoma%20is%20sudden%20intense%20exposure.
[14] Heidrich I, 05.2023: Monitoring of ctDNA in melanoma patients receiving immunotherapy, Poster presented at ISMRC 2023, Hamburg